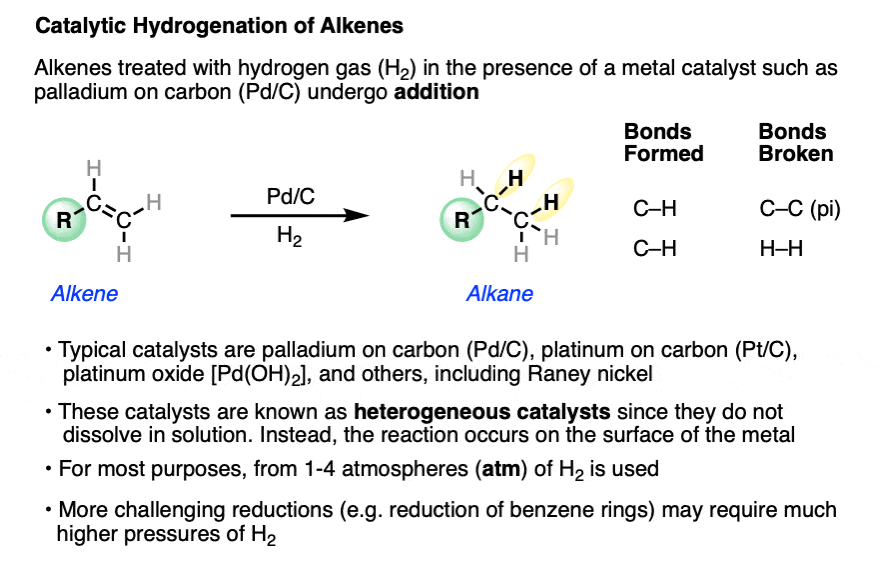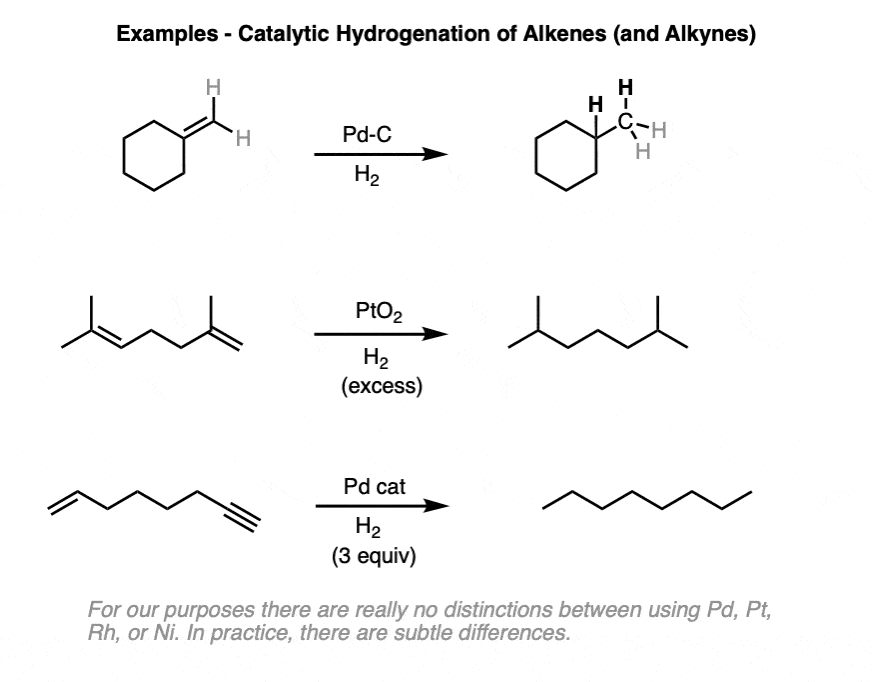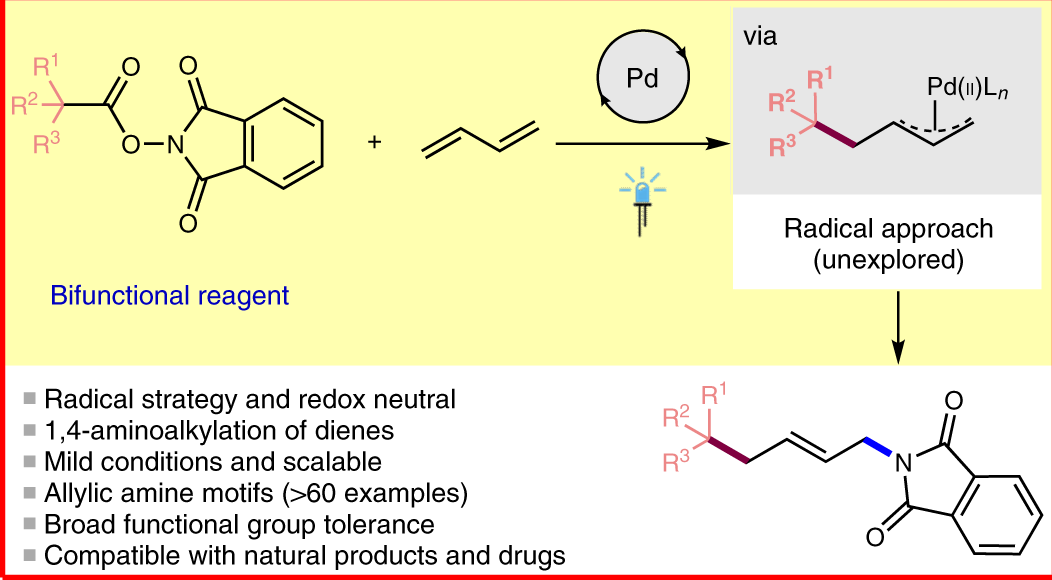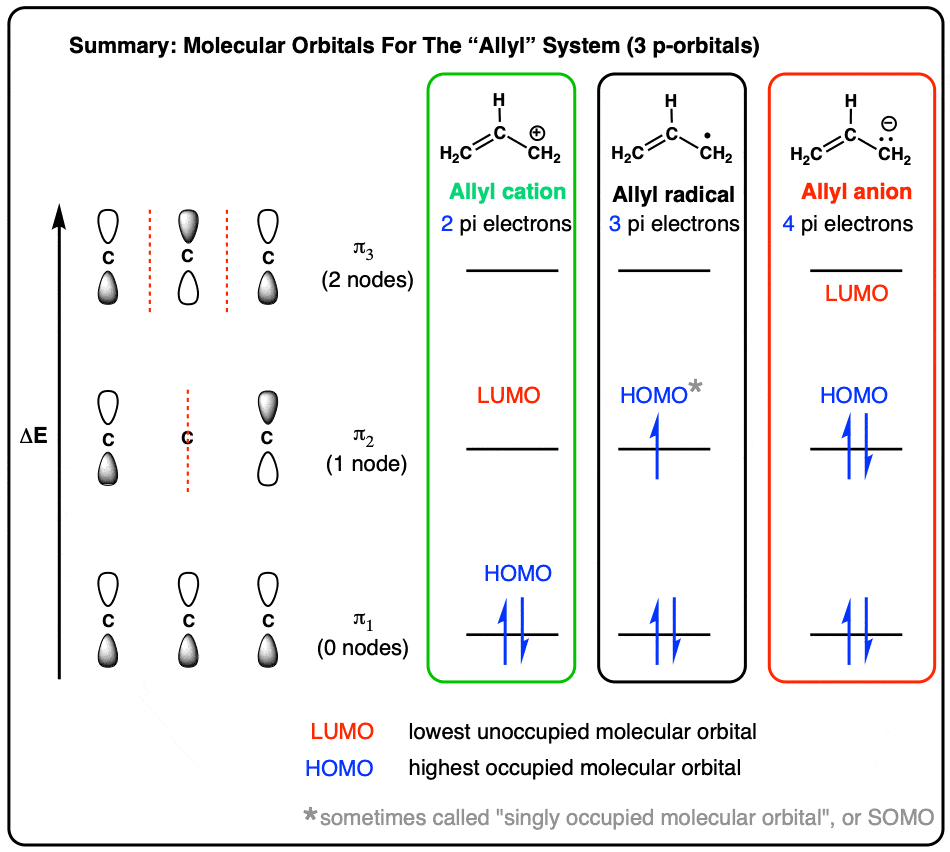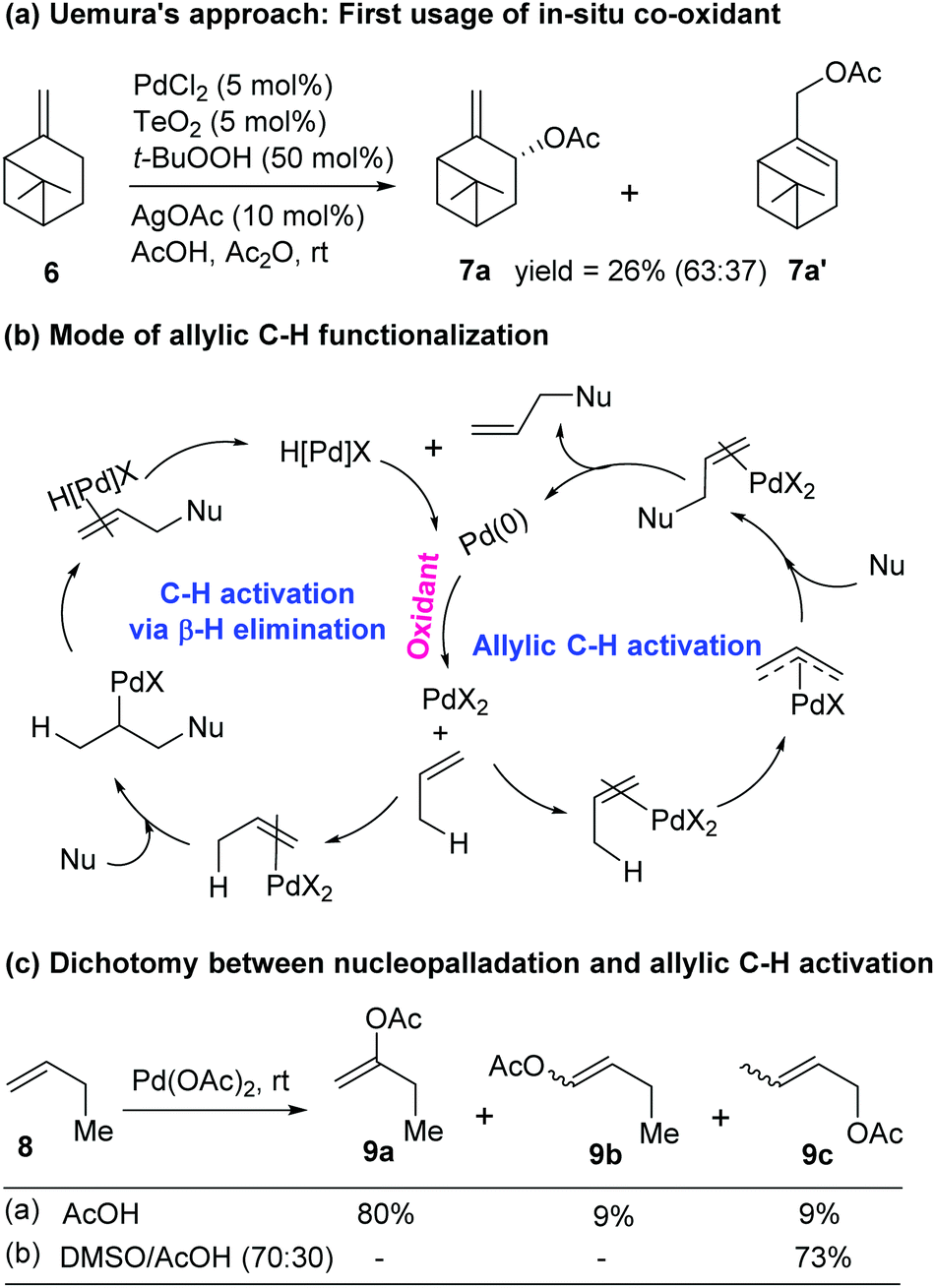
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A

Scope and Mechanism of Allylic C−H Amination of Terminal Alkenes by the Palladium/PhI(OPiv)2 Catalyst System: Insights into the Effect of Naphthoquinone | Journal of the American Chemical Society

Allylsulfones through Palladium‐Catalyzed Allylic C−H Sulfonylation of Terminal Alkenes - Chen - 2023 - European Journal of Organic Chemistry - Wiley Online Library
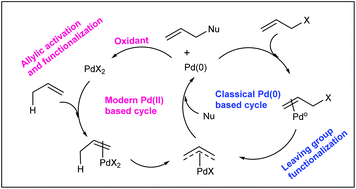
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium(II)-catalyzed alkene functionalization via nucleopalladation: stereochemical pathways and enantioselective catalytic applications. - Abstract - Europe PMC
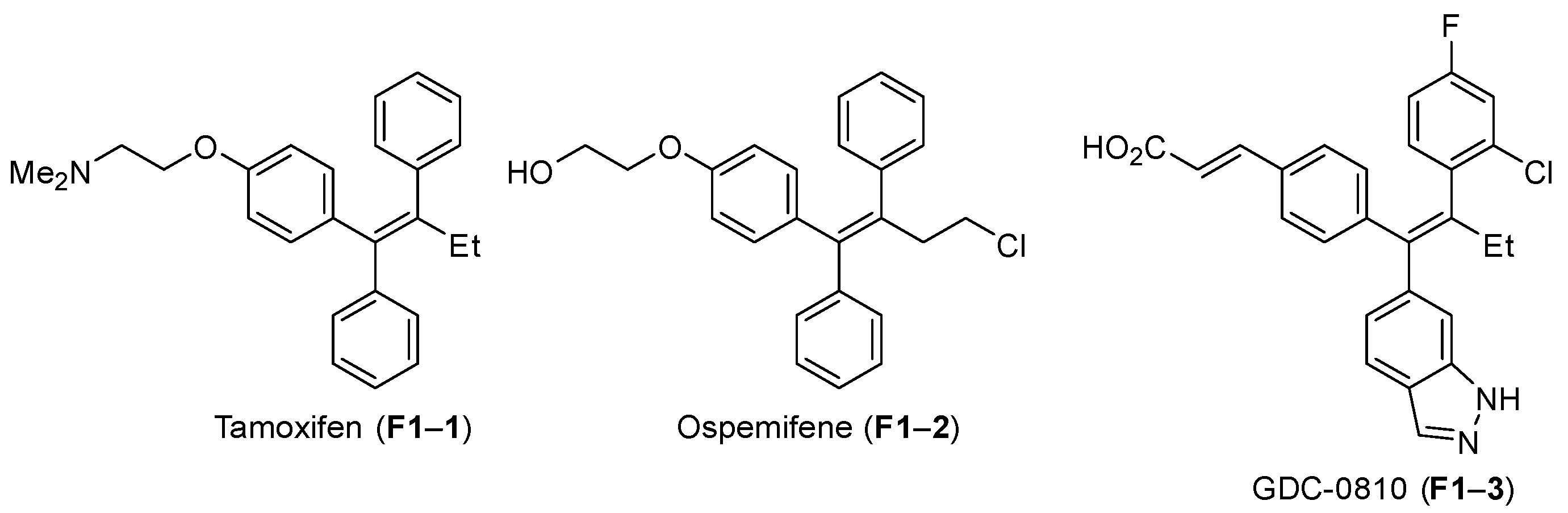
Organics | Free Full-Text | Palladium-Catalyzed Cross-Coupling Reactions of Borylated Alkenes for the Stereoselective Synthesis of Tetrasubstituted Double Bond

Allylsulfones through Palladium‐Catalyzed Allylic C−H Sulfonylation of Terminal Alkenes - Chen - 2023 - European Journal of Organic Chemistry - Wiley Online Library

O2-promoted allylic acetoxylation of alkenes: Assessment of “push” versus “pull” mechanisms and comparison between O2 and benzoquinone - ScienceDirect

Palladium-catalyzed oxidative intermolecular difunctionalization of terminal alkenes with organostannanes and molecular oxygen. - Abstract - Europe PMC
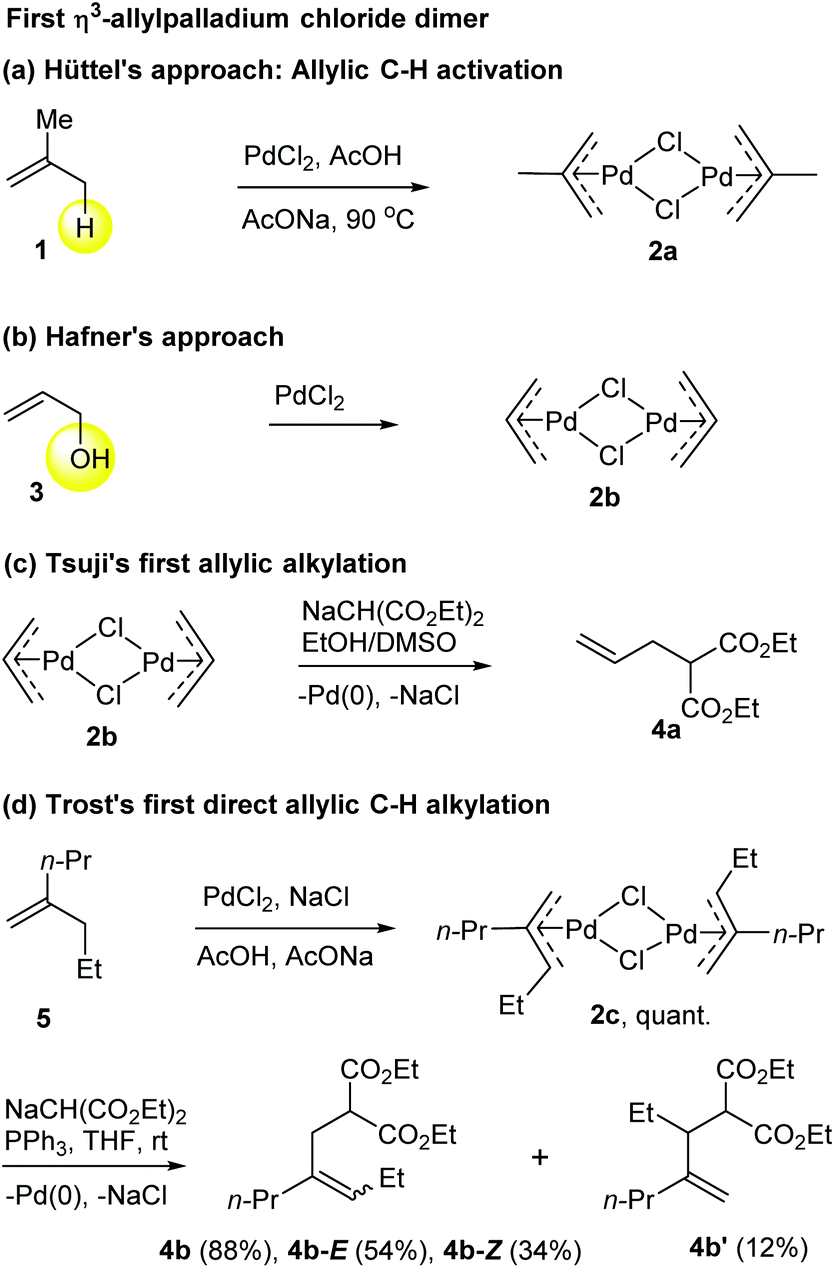
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01725A

Mechanistic Studies of Pd(II)-Catalyzed E/Z Isomerization of Unactivated Alkenes: Evidence for a Monometallic Nucleopalladation Pathway | ACS Catalysis

Generating Active “L-Pd(0)” via Neutral or Cationic π-Allylpalladium Complexes Featuring Biaryl/Bipyrazolylphosphines: Synthetic, Mechanistic, and Structure–Activity Studies in Challenging Cross-Coupling Reactions | The Journal of Organic Chemistry