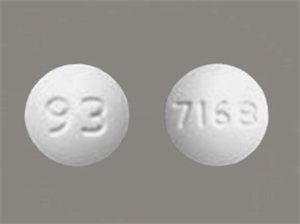
Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA

Photograph of Pack of Amlodipine 5mg Tablets. 28 tablets. Sandoz a Novartis company Stock Photo - Alamy